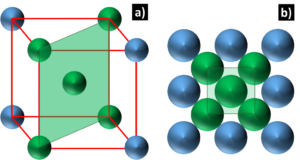
Statistics of dislocation avalanches in FCC and BCC metals: dislocation mechanisms and mean swept distances across microsample sizes and temperatures | Scientific Reports

Rolling Textures in BCC Metals: A Biaxial Stress Texture Theory and Experiments | Physics of Metals and Metallography
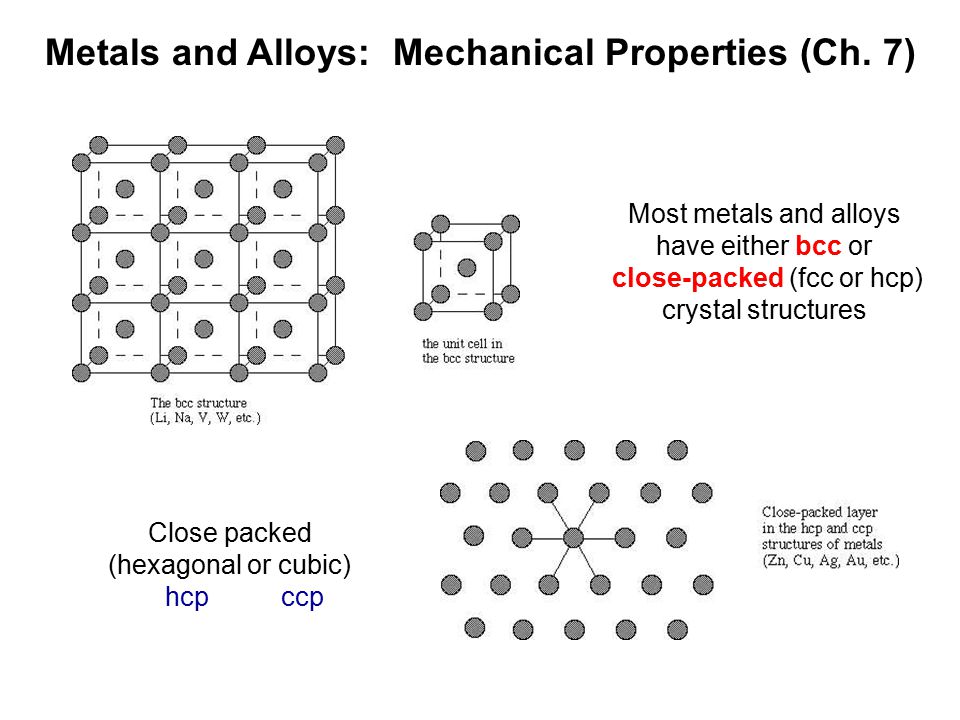
Most metals and alloys have either bcc or close-packed (fcc or hcp) crystal structures Close packed (hexagonal or cubic) hcp ccp Metals and Alloys: Mechanical. - ppt download
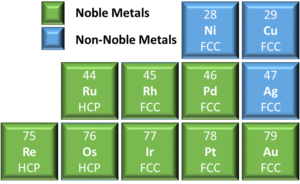
How is a crystal lattice shaped in a metal? Why is gold an FCC while titanium is a BCC? Is it the number of electrons? - Quora

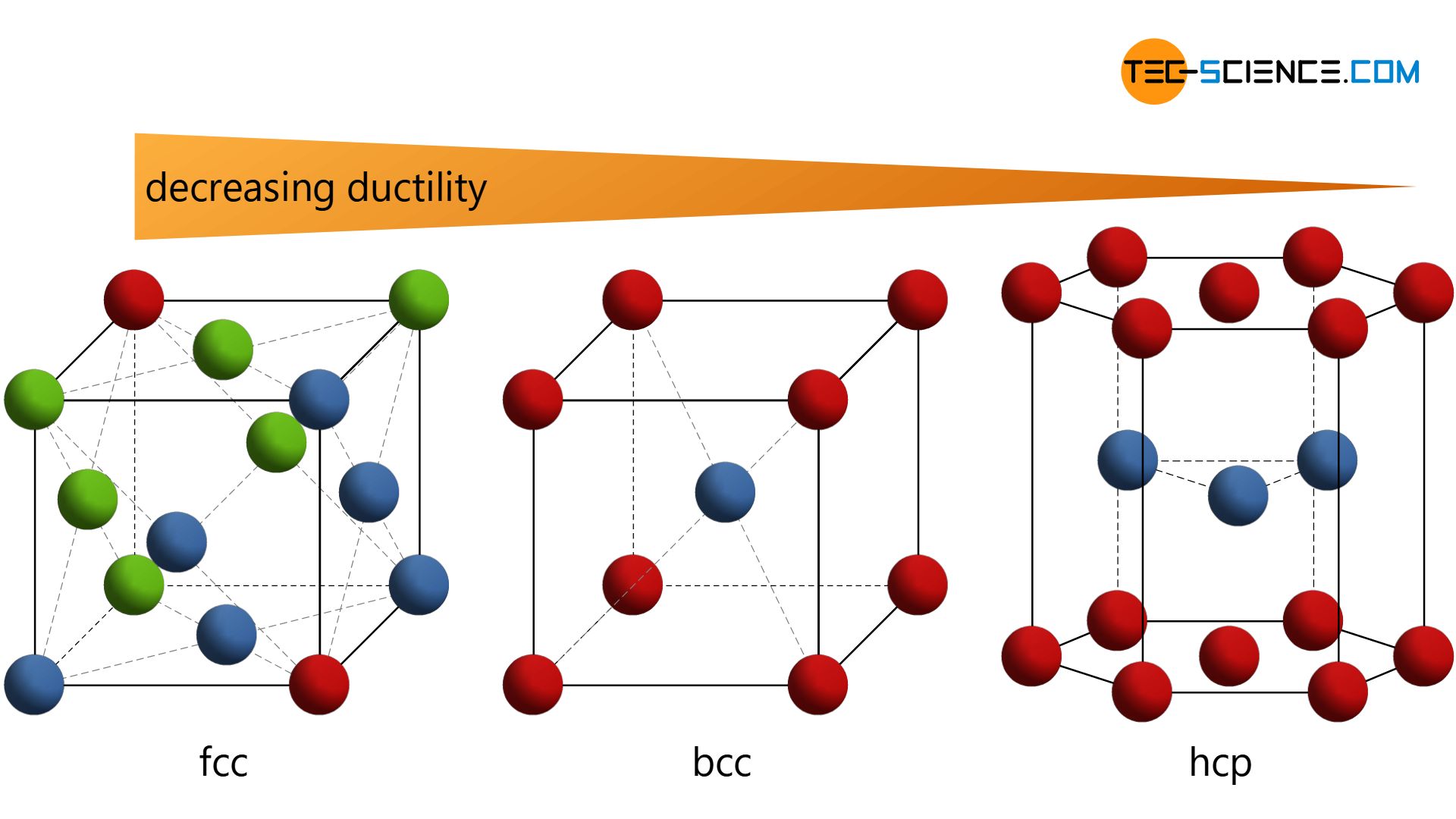
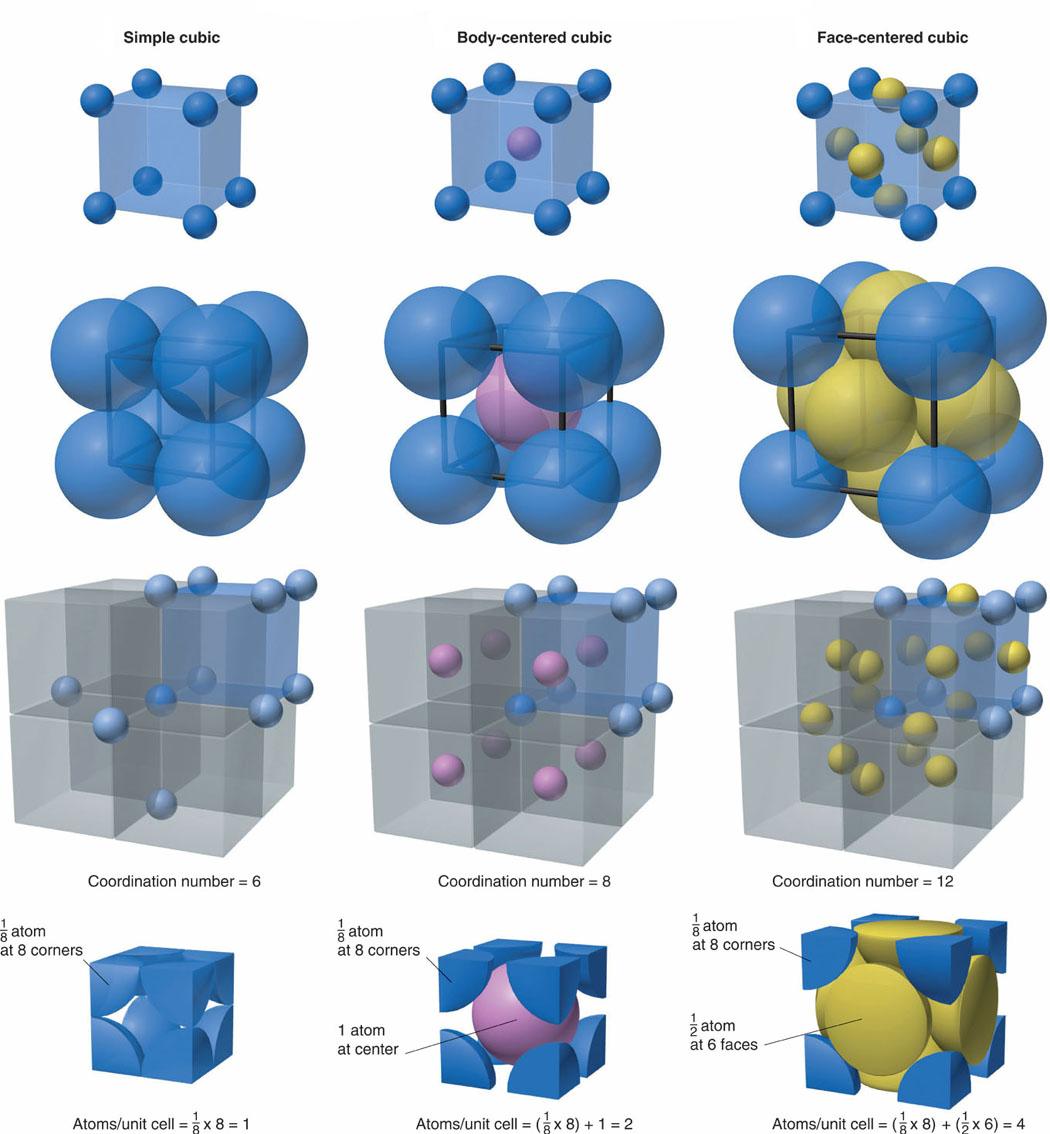
SOLVED: 6. Differentiate between Face-Centered Cubic (FCC) and Body-Centered Cubic (BCC) crystal structures. Why are FCC metals more ductile than BCC metals? 7. What is atomic packing factor (APF) and calculate the
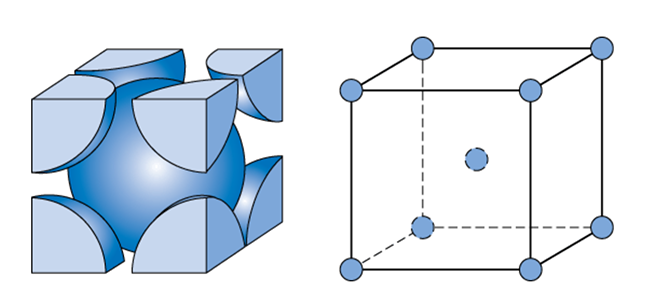
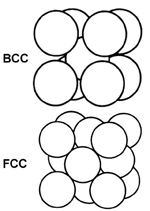
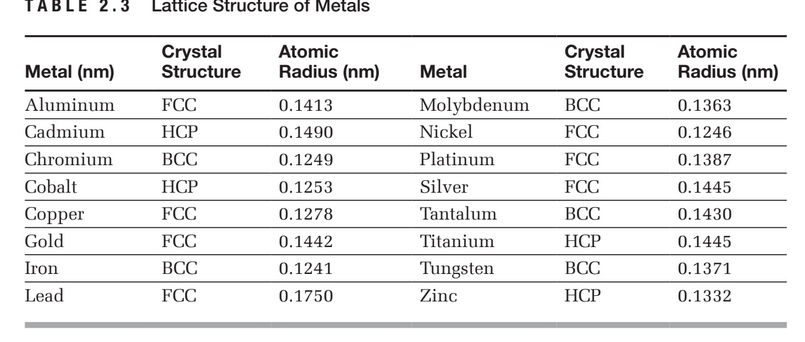
What Is the Difference Between FCC and BCC? (Crystal Structure, Properties, Interstitial Sites, and Examples) – Materials Science & Engineering
A Metal 'm' crystallizes in fcc bcc structure depending upon the temperature .The ratio of its densities in fcc bcc would be