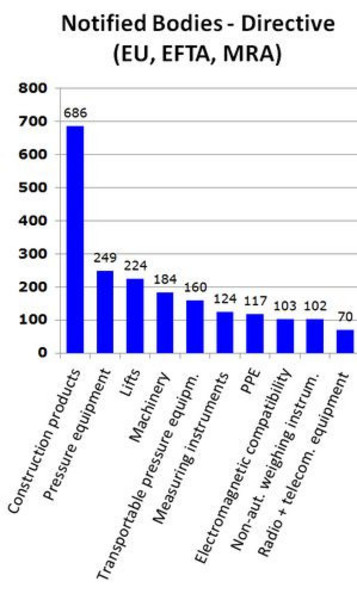
CE Mark for All Directives by EU Notified Bodies at Rs 20000/certificate in Palghar | ID: 4438471733
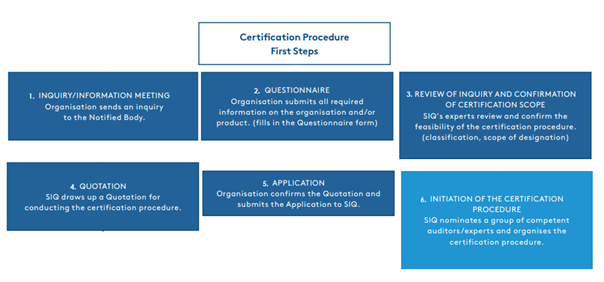
Procedure for Certification of Medical Devices in Accordance with the Regulation (EU) 2017/745 on Medical Devices (MDR) - SIQ
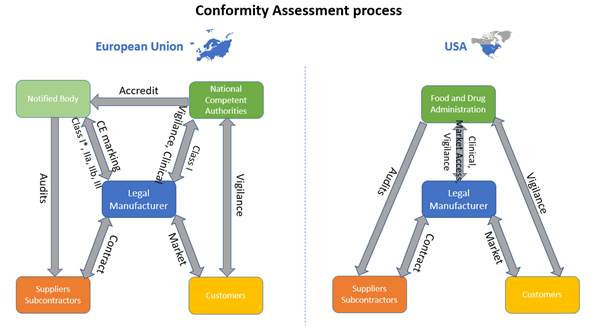
What are the principal differences between the conformity assessment process of a medical device in the USA and in the European Union? - Kvalito
![Requirements Relating to Notified Bodies for EU MDR [Video] - LearnGxP: Accredited Online Life Science Training Courses Requirements Relating to Notified Bodies for EU MDR [Video] - LearnGxP: Accredited Online Life Science Training Courses](https://learngxp.com/wp-content/uploads/2021/04/ELM-320-01-Requirements-Relating-to-Notified-Bodies-for-EU-MDR.png)