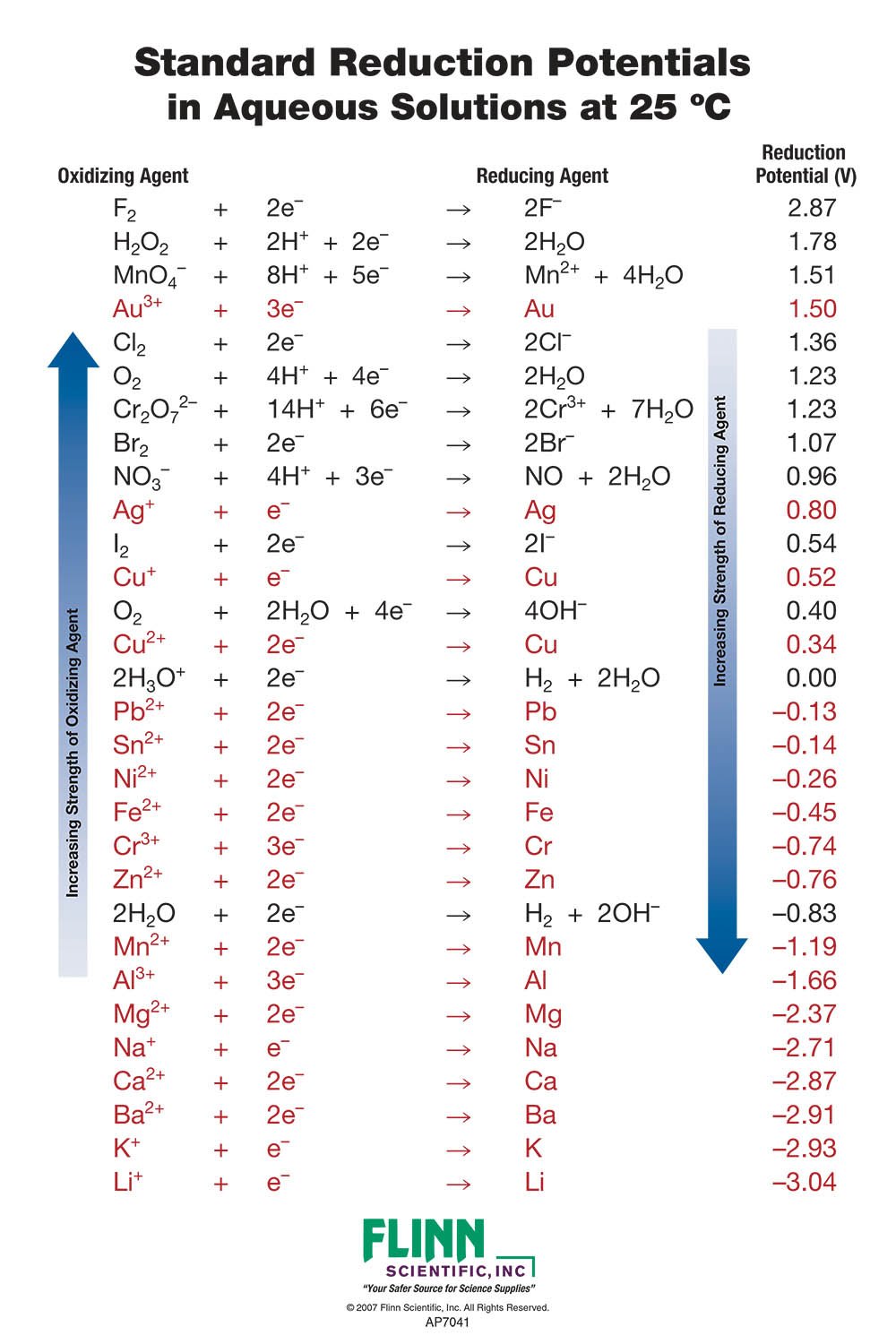
Table 1 from Building and testing correlations for the estimation of one‐electron reduction potentials of a diverse set of organic molecules | Semantic Scholar

Table 4 from Estimation of standard reduction potentials of halogen atoms and alkyl halides. | Semantic Scholar

SOLVED: TABLE 15.1 Standard Reduction Potentials of Some Biological Substances HALF-REACTION (V) O2 + 2H+ + 2e- = H2O SO4^2- + 2 H+ + 2e- = H2SO3 NO3- + 2 H+ +
Using the standard electrode potentials given in the Table, predict the reaction between the following is feasible.Ag(s) and Fe^{3+}(aq)

Using the standard electrode potentials given in the table, predict the reaction between the following is possible.Fe^{3+}(aq) and I^{-}(aq)



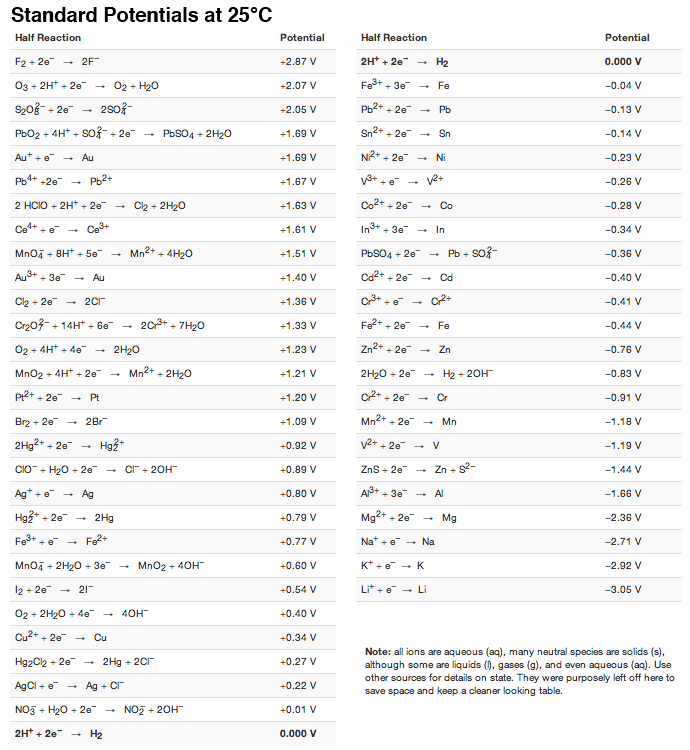
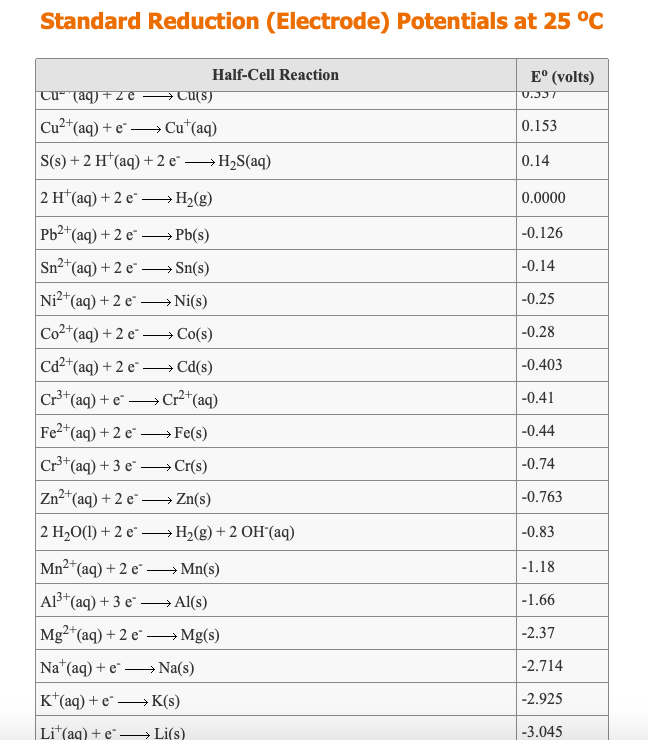

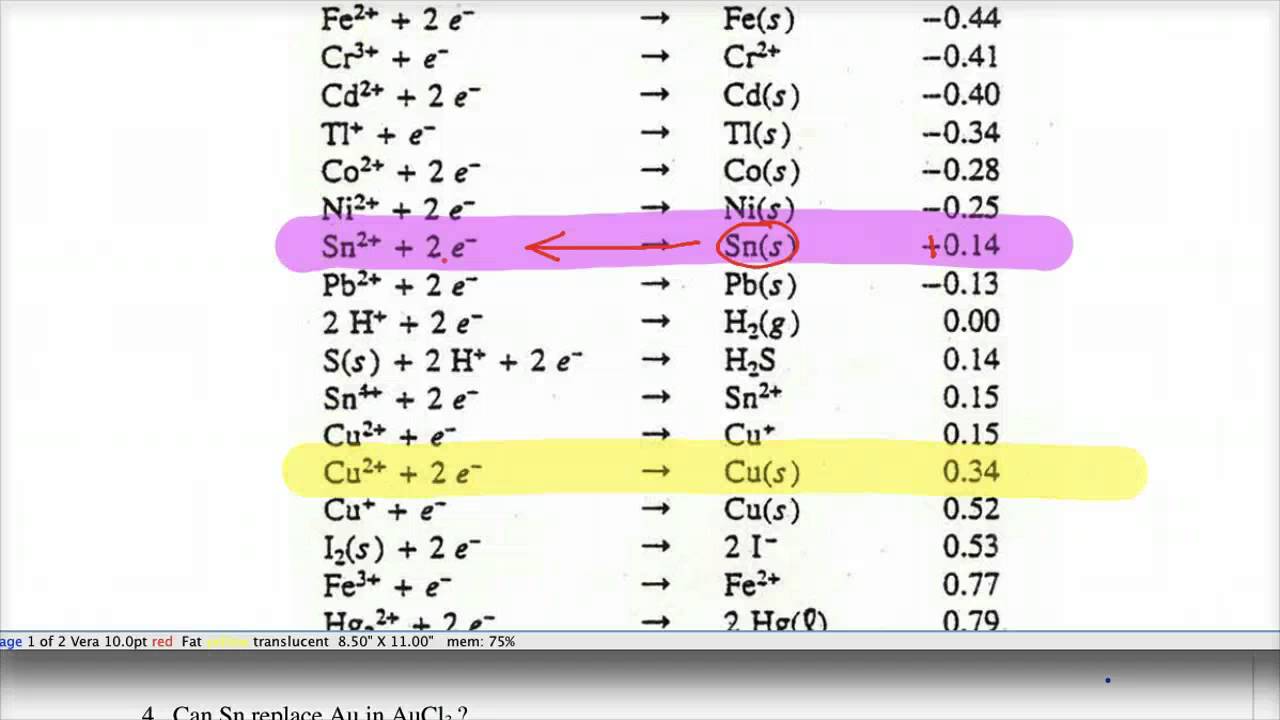

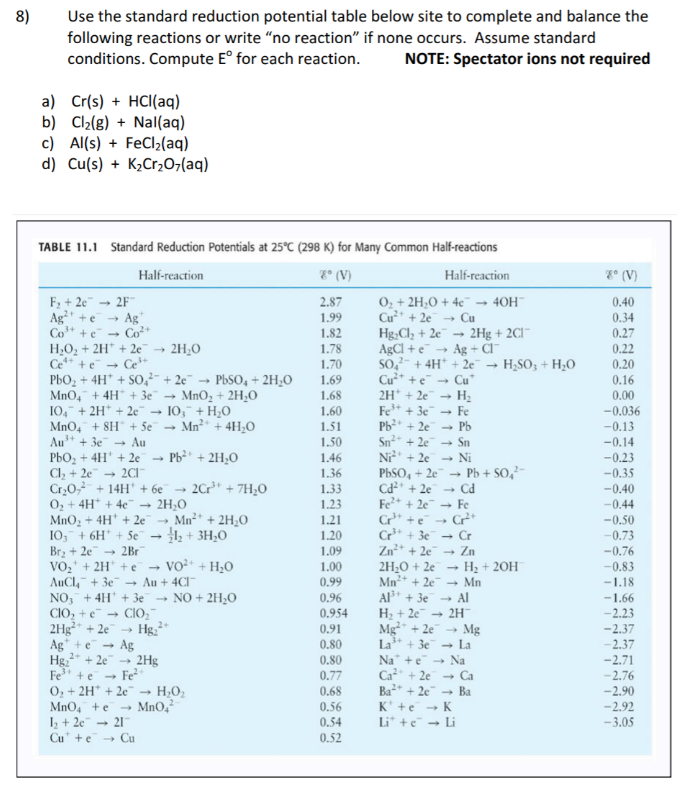


![Standard reduction potentials at 298°K. [24] | Download Table Standard reduction potentials at 298°K. [24] | Download Table](https://www.researchgate.net/publication/316026333/figure/tbl2/AS:650784626708491@1532170554986/Standard-reduction-potentials-at-298K-24.png)