![PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b0d76592f078aeeb6e66f500b45a1f101c6fe150/5-Table1-1.png)
PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar
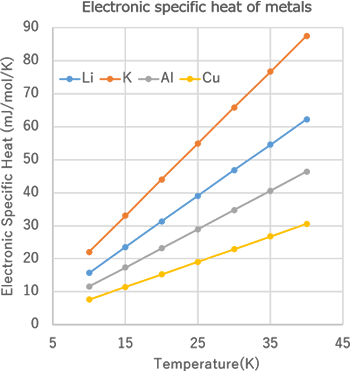
Electronic specific heat analysis of metals - J-OCTA Case Studies | CAE Solutions - JSOL Corporation
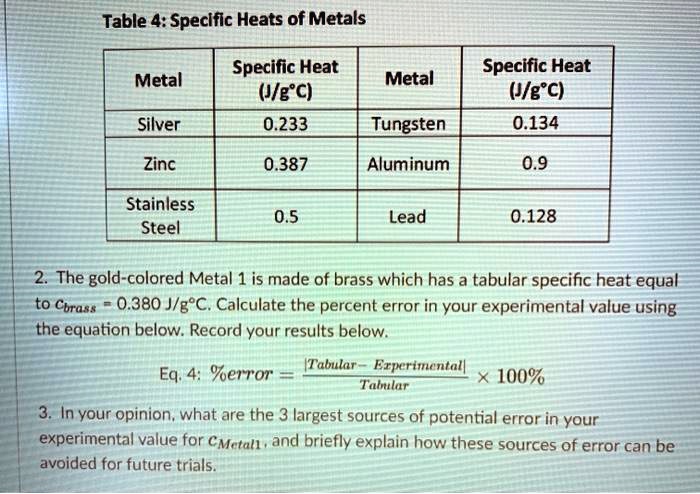
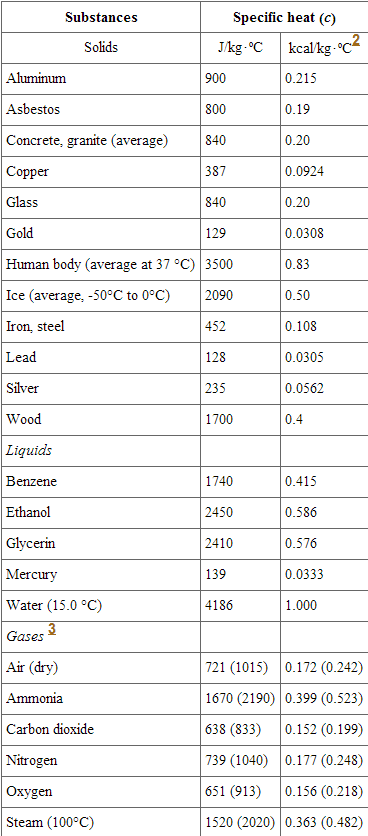
SOLVED: Table 4: Specific Heats of Metals Metal Specific Heat (J/g°C) Tungsten 0.233 Silver 0.134 Zinc 0.387 Aluminum 0.9 Stainless Steel 0.5 Lead 0.128

PDF) A new correlation for the specific heat of metals, metal oxides and metal fluorides as a function of temperature
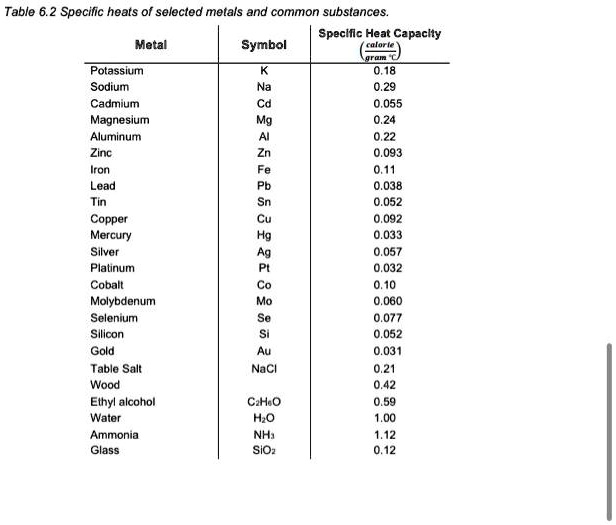
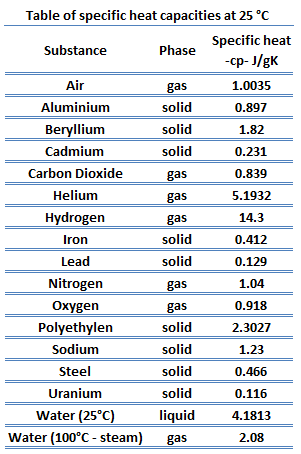
SOLVED: Table 6.2: Specific heats of selected metals and common substances Metal Symbol Specific Heat Capacity Potassium K 0.18 Sodium Na 0.55 Cadmium Cd 0.93 Magnesium Mg 0.11 Aluminum Al 0.038 Zinc
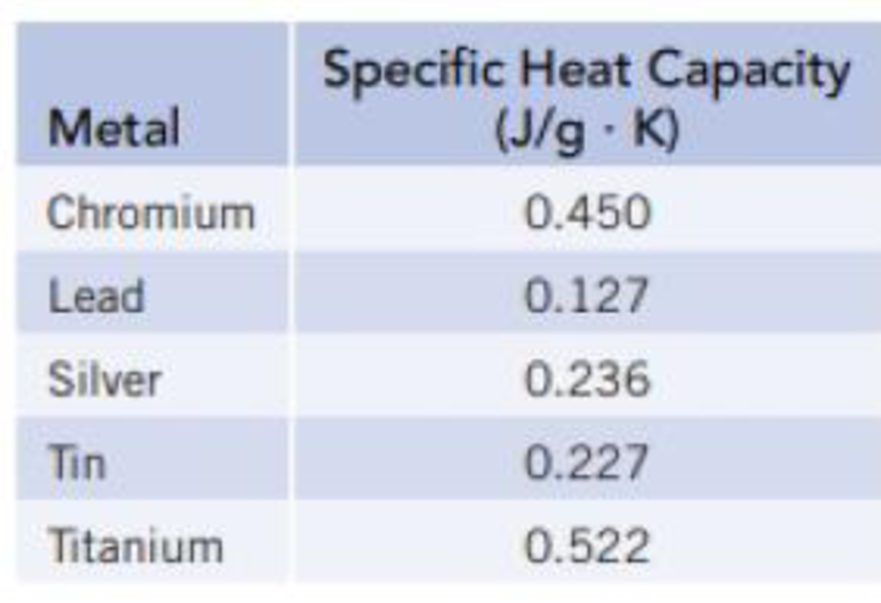
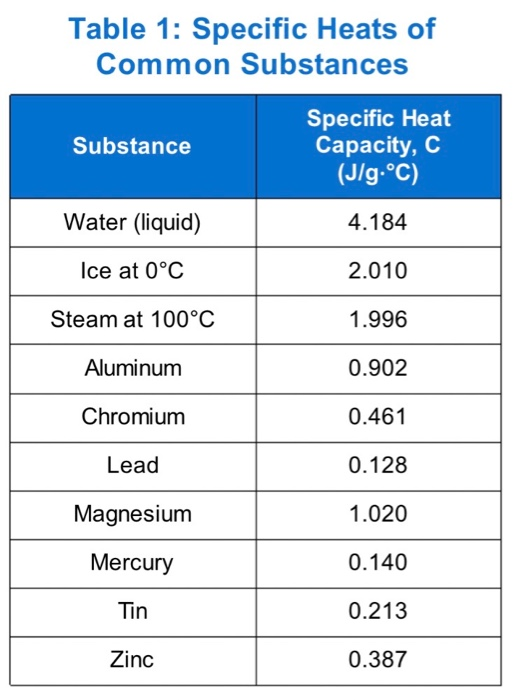
Prepare a graph of specific heat capacities for metals versus their atomic weights. Combine the data in Figure 5.4 and the values in the following table. What is the relationship between specific
![PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b0d76592f078aeeb6e66f500b45a1f101c6fe150/6-Table3-1.png)
PDF] A NEW CORRELATION FOR THE SPECIFIC HEAT OF METALS, METAL OXIDES AND METAL FLUORIDES AS A FUNCTION OF TEMPERATURE | Semantic Scholar
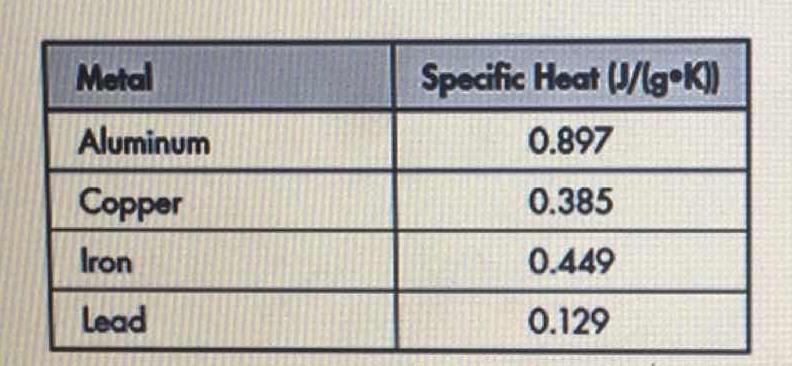
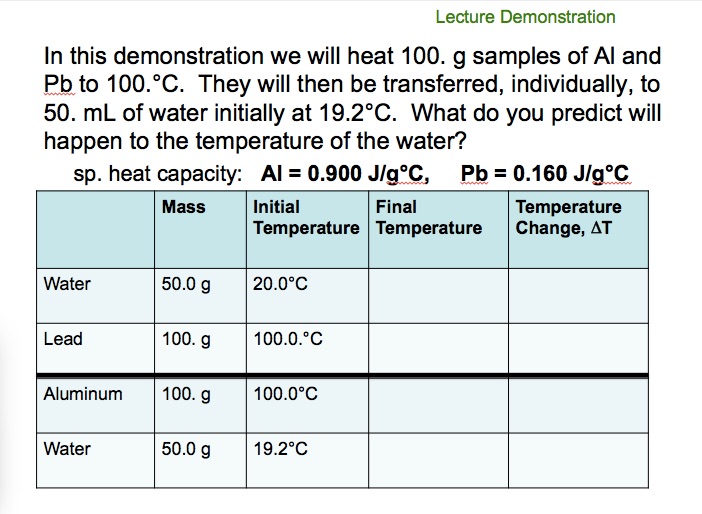
The table below shows the specific heats of several metals. The temperature of a 15-g sample of an unknown metal increases from 20.0 C to 30.0 C when it absorbs 67.5 J